Finally, add together the total mass of each element to get the molar mass of O: 15.9994 g/mol 15.9994 g/mol. Since there are 2 hydrogens in the molecule, the total weight of hydrogen in water is 2 times 1. Calculate the molar mass of PO4 in grams per mole or search for a chemical formula or substance. For hydrogen, the atomic weight is 1.00794, and for oxygen it is 15.9994. We then take the atomic weight of the atoms from the periodic table. The molecular mass of water is 18.0 amu, and the molar mass is 18.0 g/mol. We can calculate the molecular weight of a substance using its chemical formula and the periodic table. For example, we can say that the mass of Cu is 63.55 amu or 63.55 g/mol. You will need to refer to a periodic table for proton values. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in O: Molar Mass (g/mol) O (Oxygen) 1 × 15.9994 15.9994. Numerically, the molar mass is equal to the atomic mass of a given atom or a molecule, so we can look up the molar mass of an element in the periodic table. In this notation, the atomic number is not included. Symbol-mass format for the above atom would be written as Cr-52. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. The given mass of K (4. Both the atomic number and mass are written to the left of the chemical symbol. Referring to the periodic table, the atomic mass of K is 39.10 amu, and so its molar mass is 39.10 g/mol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#).\nonumber \] Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: The atomic mass of Copper is 63.55 atomic mass units. Atomic mass is usually listed below the symbol for that element. If your sample is made of one element, like copper, locate the atomic mass on the periodic table. Find a periodic table of elements to find the molar mass of your sample. png file can be downloaded here (2017 values for all 118 elements). Step 1: Find the Molar Mass of the Formula. This table can be used for computer and mobile device wallpapers. 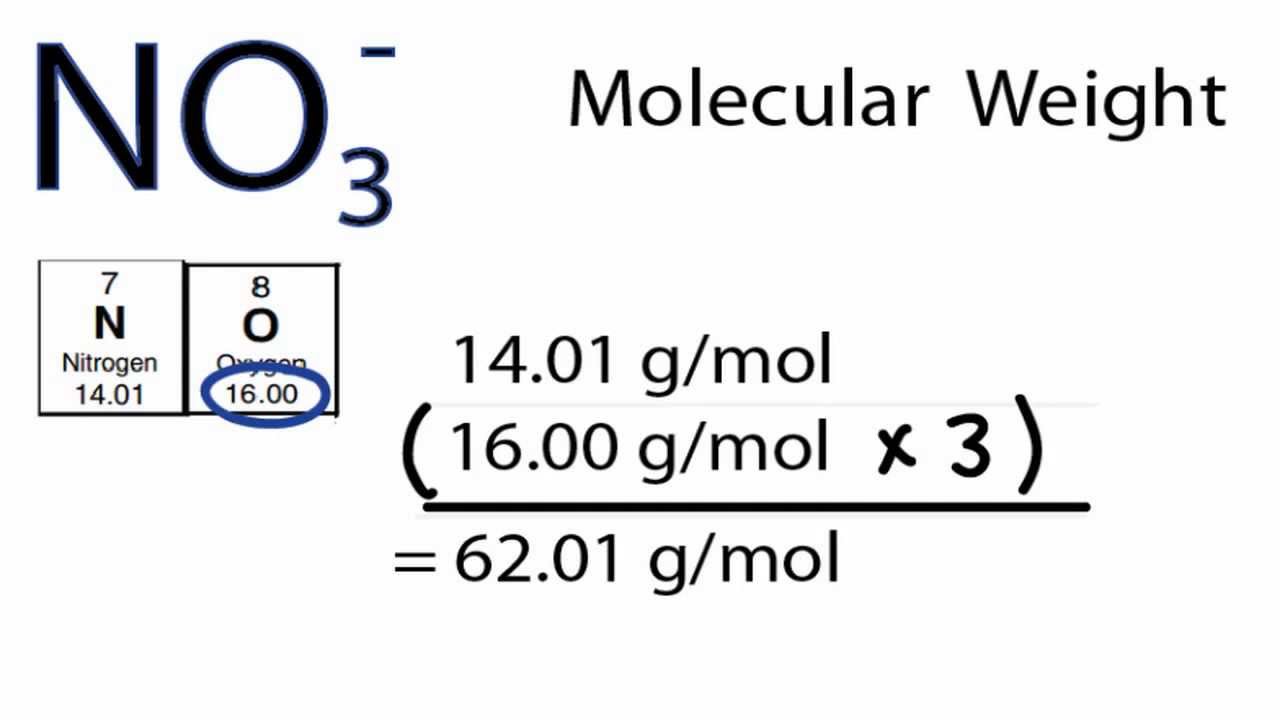
This table can be obtained in PDF format here. There you can find the metals, semi-conductor (s), non-metal (s), inert noble gas (ses), Halogens, Lanthanoides. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. I prefer to use tables with whole number atomic masses, but that is just personal preference. The lightest chemical element is Hydrogen and the heaviest is Hassium. Find a periodic table and look at the atomic mass of each atom in the molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
